Forever Chemicals, Forever Vaults: Why Cancer Sequesters and Stores PFAS
PFAS are modern industrial toxins that our bodies struggle to break down, which is why the body recruits cancer tumors to store these forever chemicals.
Welcome to my blog!
Not all breakthroughs in synthetic chemistry are good for us. It turns out that if you take a nice, well-behaved organic molecule and replace all the hydrogen atoms with fluorine atoms, you get an evil monster that’s called a “forever chemical”.
Forever chemicals have their name for a very good reason: they never break down. They are also known as PFAS — per- and polyfluoroalkyl substances. These synthetic molecules, used for decades in non-stick cookware, waterproof clothing, food packaging, cosmetics, and firefighting foams, have now contaminated the drinking water, soil, and blood of virtually every person on the planet.
And they are accumulating.
RFK Jr. has publicly advocated for ending PFAS production in America. While governments and researchers scramble to measure the damage and debate regulations, one critical question remains largely ignored: What is the body actually doing with these persistent toxins?
Under Toxin Sequestration Theory (TST), the answer becomes clear. When the body faces a toxin it cannot efficiently break down or excrete — especially one that generates chronic oxidative stress — it eventually builds secondary storage vaults. In the case of PFAS, those vaults are tumors. Hence PFAS add to our growing list of carcinogens, shown in the figure below.

This post explores how PFAS act as a textbook example of modern toxic overload, why they drive cancer formation in specific organs, and how cancer cells actively sequester and store these forever chemicals.
What Are PFAS and Where Do We Encounter Them?
PFAS are a large family of synthetic chemicals characterized by extremely strong carbon-fluorine bonds. These bonds make PFAS nearly indestructible — they resist heat, water, oil, and degradation for decades or even centuries. This is why they’re called “forever chemicals.”
Since the 1950s, PFAS have been widely used in:
Non-stick cookware (Teflon)
Water- and stain-resistant clothing and carpets
Food packaging (grease-proof wrappers, microwave popcorn bags)
Cosmetics and personal care products
Firefighting foams used at military bases and airports
Industrial processes and pesticides
Today, PFAS are ubiquitous. They contaminate public drinking water supplies, soil, and food chains. Studies show that nearly every person in industrialized countries has detectable levels of PFAS in their blood. Once inside the body, they bind strongly to proteins (especially albumin) and accumulate in organs with high lipid content or filtration roles — particularly the kidneys, liver, and testes.
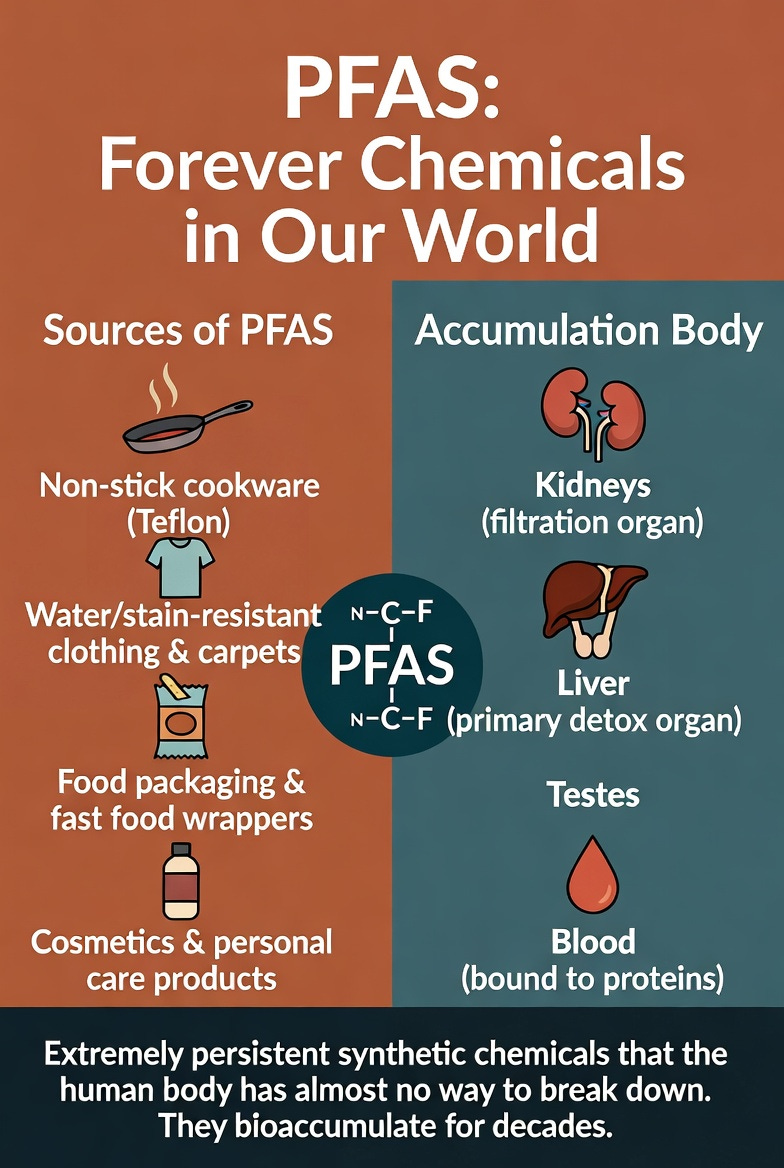
Unlike many toxins we’ve discussed (such as oxalates or seed oils), PFAS are not easily metabolized. The human body has almost no natural enzymes capable of breaking their carbon-fluorine backbone. This extreme persistence turns them into a long-term toxic burden that steadily increases oxidative stress over time.
PFAS as a Toxin: The ROS Connection
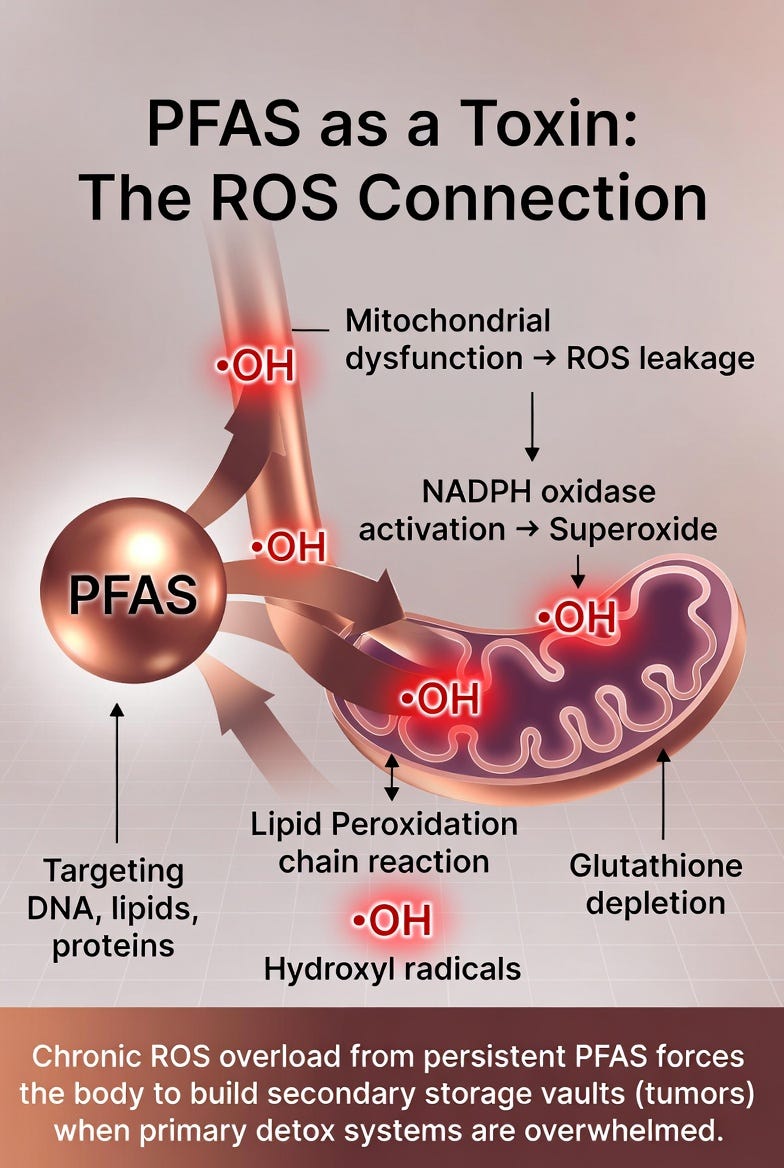
PFAS don’t just sit quietly in the body — they actively generate oxidative stress. Despite their chemical stability, these forever chemicals disrupt cellular redox balance in several ways:
They impair mitochondrial function, leading to increased leakage of reactive oxygen species (ROS).
They activate NADPH oxidase, an enzyme that produces superoxide radicals.
They trigger lipid peroxidation in cell membranes, creating a chain reaction of oxidative damage.
They deplete key antioxidant defenses, particularly glutathione.
The result is chronic, low-grade ROS overload. Unlike a one-time toxin exposure, PFAS persist for years or decades, continuously feeding this oxidative fire. This sustained redox stress damages proteins, lipids, and DNA over time — exactly the kind of cellular chaos that Toxin Sequestration Theory predicts will eventually force the body to build protective storage vaults (tumors).
What makes PFAS especially problematic is their indestructibility. While the body has enzymes and pathways to handle glucose, seed oils, iron, copper, and even microplastics to some degree, it has almost no effective way to metabolize or break down PFAS. When primary detox organs (liver and kidneys) become overwhelmed, the body is left with a growing toxic burden it cannot eliminate.
This is where tumors enter the picture — as secondary containment systems designed to isolate and manage persistent toxins the body cannot clear.
The Body’s Normal Defense Systems (and Why They Fail)
The human body is well-equipped to handle many toxins — but PFAS expose a critical weakness in our detoxification machinery.
Under normal conditions, the liver attempts to conjugate and package toxins for elimination, while the kidneys filter and excrete them through urine. The body also uses glutathione and other antioxidants to neutralize ROS, and bile flow to remove fat-soluble compounds.
PFAS break this system.
Because of their ultra-stable carbon-fluorine bonds, PFAS resist almost all metabolic breakdown. The liver cannot effectively conjugate them, and the kidneys excrete them very poorly. Instead, PFAS bind tightly to blood proteins (especially albumin) and accumulate in tissues with high blood flow or lipid content — particularly the kidneys, liver, and testes.
Over years of exposure, this leads to a slow but steady buildup. Primary detox organs become overwhelmed, antioxidant reserves are chronically depleted, and systemic ROS levels remain elevated. The body is left with a growing toxic load it has no efficient way to eliminate.
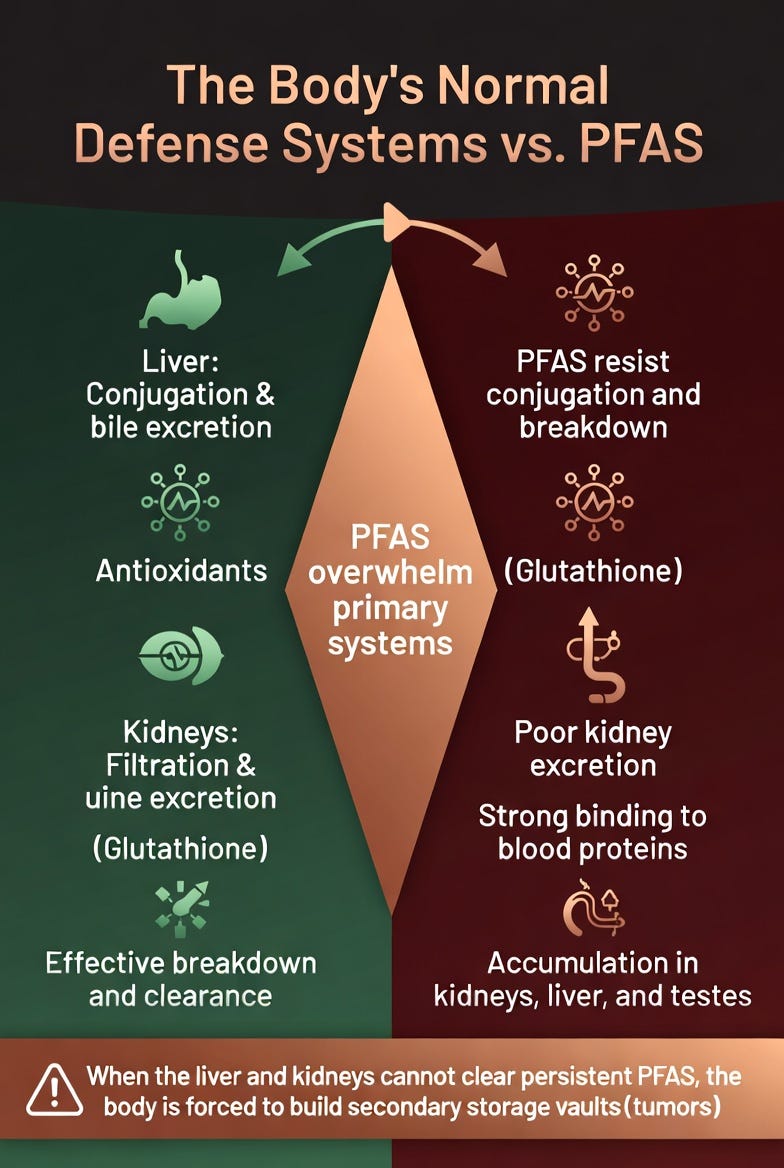
This is the exact scenario where Toxin Sequestration Theory (TST) predicts the body will activate its backup strategy: building secondary containment sites — tumors — to isolate and store the persistent chemicals.
Cancer Sequesters PFAS: The Body’s Final-Line Defense
In TST, tumors function as adaptive, specialized compartments the body creates to isolate persistent toxins that overwhelm the liver, kidneys, and other primary clearance systems. We’ve seen this pattern repeatedly: tumors rapidly import and burn glucose to limit systemic oxidative stress; they convert fructose into lipids as a “second liver”; they pull in glutamine to detoxify ammonia; and they store oxidized seed oils in intracellular lipid droplets. PFAS — the so-called “forever chemicals” — follow the same logic with remarkable precision.
PFAS are extraordinarily stable, protein-loving molecules that resist breakdown and excretion. They bind tightly to serum proteins like albumin and globulins, circulate widely, and accumulate preferentially in the liver, kidneys, lungs, and testes — exactly the organs with the strongest links to PFAS-associated cancers. Their half-lives in humans range from years to decades, creating a chronic internal burden that standard detox pathways cannot fully resolve.
When PFAS load becomes excessive, the body escalates to the same sequestration strategy used for other toxins: it forms or expands tumor microenvironments that actively pull these chemicals out of circulation and isolate them. This reframes PFAS-linked “cancer” not as random malignancy but as a symptom of overwhelming toxic overload — the body’s attempt to protect vital organs by creating a dedicated containment site.
Evidence and Mechanisms: How Tumors Act as PFAS Sinks
Direct evidence supports this protective sequestration role:
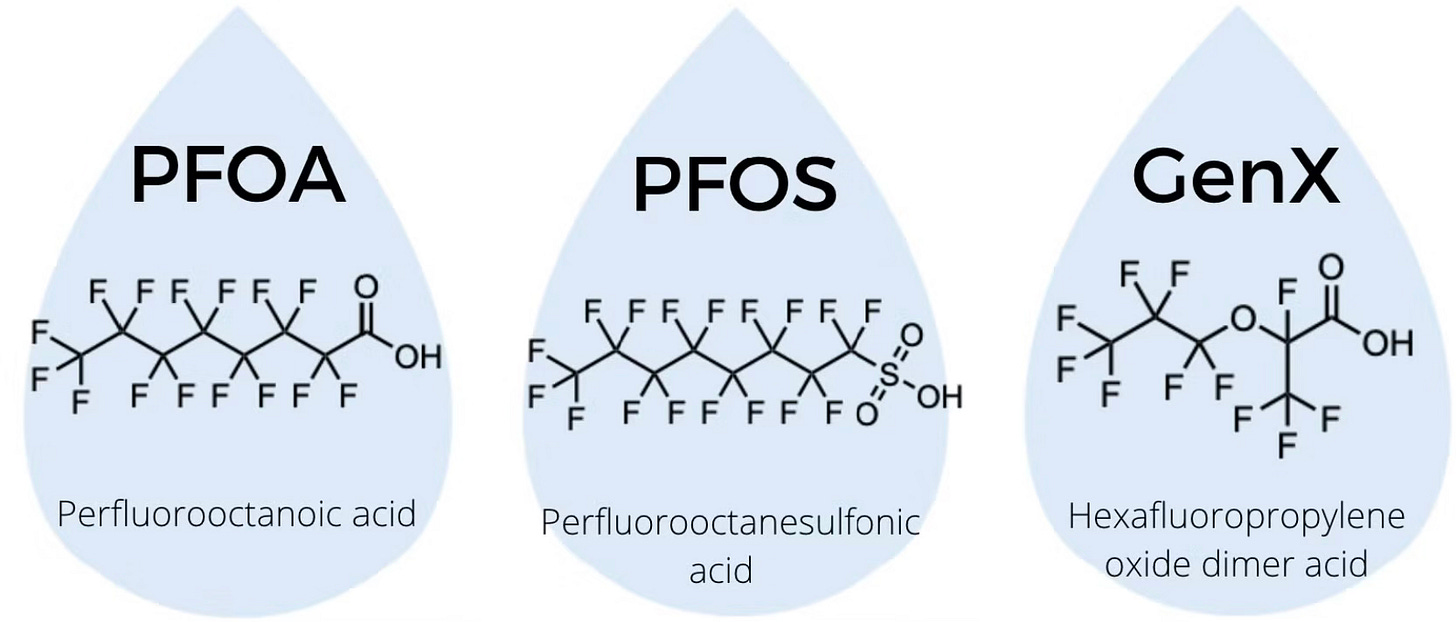
Studies of lung adenocarcinoma patients show significantly higher PFAS levels (especially PFOA and PFOS) in both serum and tumor tissues compared to healthy controls. PFOA, in particular, constitutes a large fraction of detected PFAS in cancerous lung tissue, indicating active accumulation inside the tumor.
In hepatobiliary and gastrointestinal cancers, PFAS analytes are detectable in a substantial portion of tumor samples, often correlating with serum levels.
Strong epidemiological ties exist between PFAS exposure and cancers in precisely the organs where these chemicals concentrate most: kidney, testicular, liver, and lung.
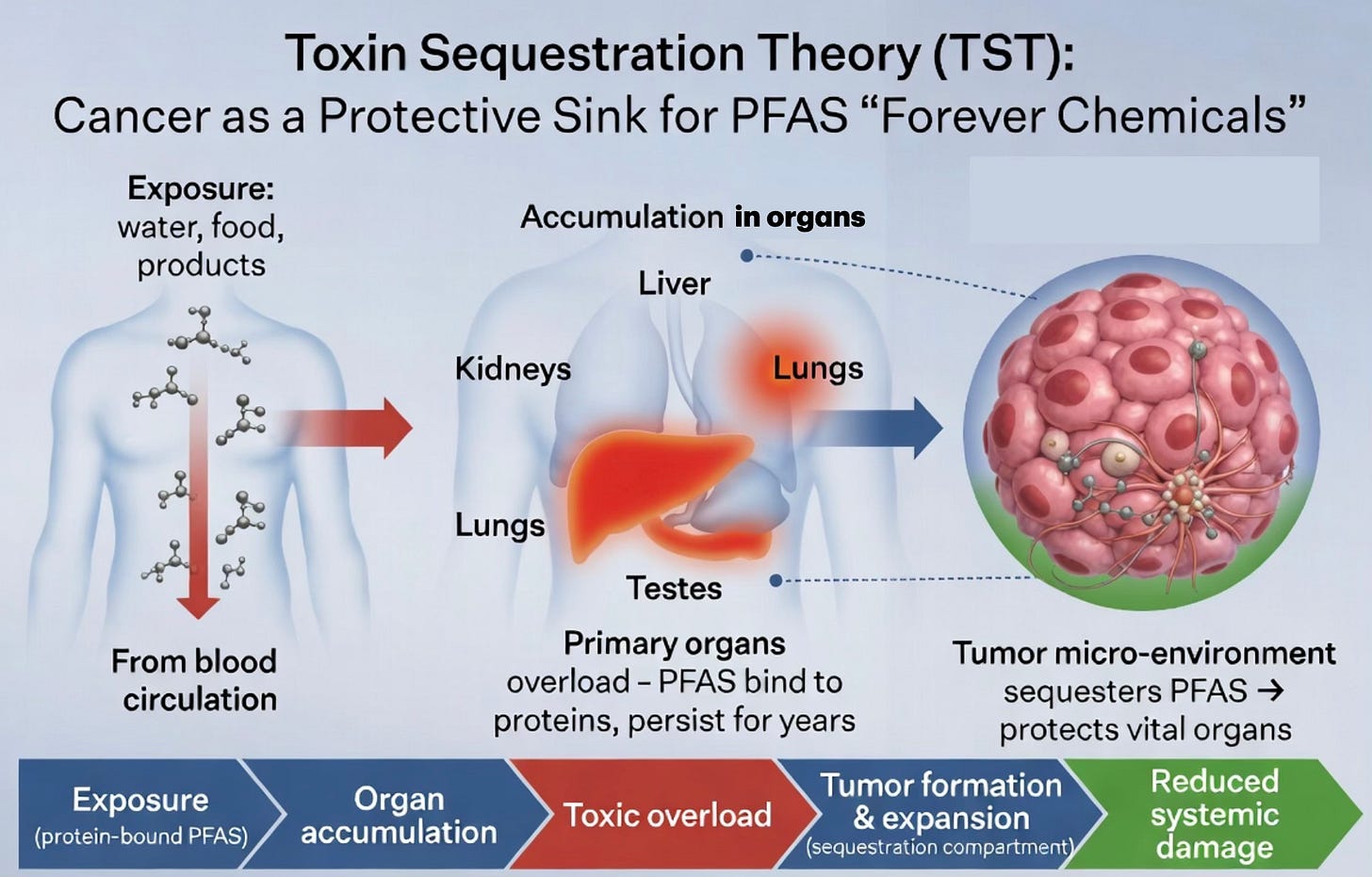
Tumors appear to achieve this isolation through mechanisms that align with their established biology and our prior TST work:
Protein-rich binding environment — Tumors express high levels of proteins and create localized conditions that enhance PFAS retention, leveraging the same proteinophilic affinity that makes these chemicals so persistent in blood.
Dysregulated lipid and metabolic dynamics — Even though PFAS are primarily protein-bound, the altered lipid metabolism and lipid droplet formation we discussed in the seed-oil post can still help partition and sequester them.
Immunosuppressive niche — The tumor locally dampens immune activity, preventing aggressive clearance that might redistribute PFAS back into circulation and damage distant organs.
High metabolic throughput — The rapid uptake of glucose, fructose, and glutamine documented earlier creates a powerful “sink” capable of also binding, conjugating, or isolating PFAS where possible.
This protective function explains otherwise puzzling observations: why PFAS exposure so consistently triggers tumors in accumulation-prone tissues, why serum PFAS levels track with cancer risk, and why reducing exposure sometimes improves outcomes — the sequestration pressure eases as the toxin burden drops.
Experimental data showing PFAS increasing tumor growth or cell migration (e.g., in testicular or colorectal models) fit perfectly here: in TST, this reflects the body ramping up the sequestration compartment to handle the load, not a simple “carcinogenic” effect.
How Cancer Cells Actively Pull PFAS Out of Body Fluids
Cancer cells don’t just passively absorb PFAS — they actively pull them out of blood and interstitial fluid as part of the body’s protective sequestration program.
PFAS circulate in blood tightly bound to albumin and other serum proteins. Tumors, being highly vascular and metabolically aggressive, exploit this by upregulating key transporters such as Organic Anion Transporting Polypeptides (OATPs) and Fatty Acid-Binding Proteins (FABPs). These same transporters that cancer cells use to import nutrients also shuttle PFAS across the cell membrane. Once inside, their amphipathic nature allows them to partition into membranes and concentrate in the expanded lipid droplets we discussed earlier.
In TST terms, this is not random uptake — it is a deliberate sink. The tumor microenvironment creates strong concentration gradients through high protein content, rapid lipid synthesis, and altered pH, efficiently drawing PFAS out of circulation and isolating them. Just as tumors import glucose, fructose, and glutamine at accelerated rates, they pull in and sequester “forever chemicals” to shield the rest of the body.
Cancer Cells store PFAS in Lipid Droplets
Tumors readily form and expand lipid droplets (LDs) — the same protective organelles we highlighted in the seed-oil post for sequestering oxidized polyunsaturated fats.
PFAS exposure strongly promotes lipid dysregulation and LD accumulation, especially via increased triglyceride synthesis in response to PFOA and PFOS. Although primarily protein-bound, PFAS can partition at lipid-water interfaces and co-accumulate inside these droplets.
In the TST framework, cancer cells use LDs as a secondary sequestration compartment: they isolate PFAS (alongside toxic lipids) away from circulation and sensitive organs like the liver and kidneys. The tumor’s high-metabolic, immunosuppressive niche stabilizes this storage, preventing systemic release.
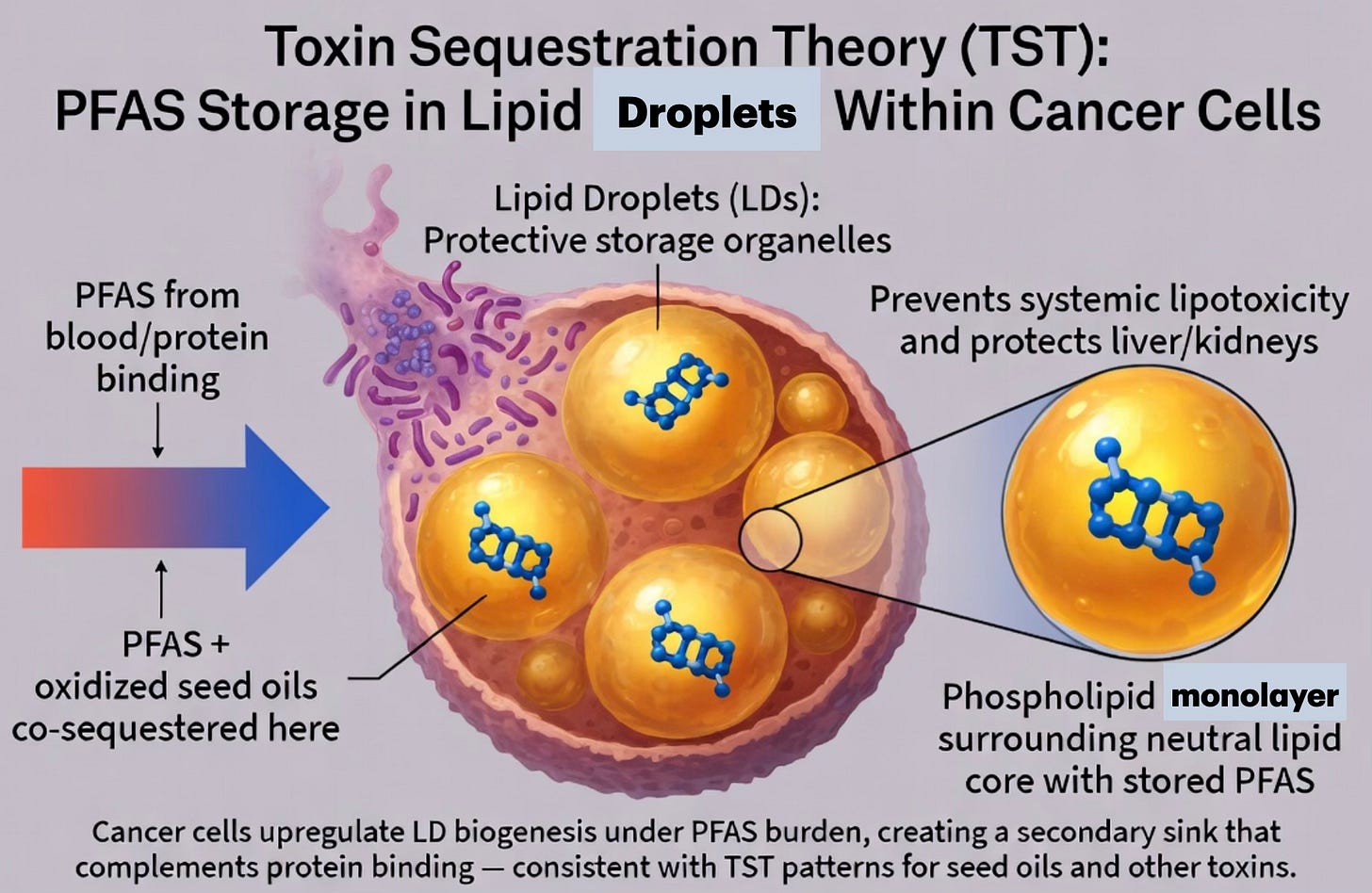
This dual strategy — protein binding plus lipid droplet storage — explains elevated PFAS levels inside tumors and why exposure drives tumor growth: the body is actively ramping up LD-rich sequestration sites to contain the persistent “forever chemical” load.
PFAS-to-Organ Mapping: Why Tumors Form Precisely Where These “Forever Chemicals” Accumulate
One of the clearest predictions of the Toxin Sequestration Theory is the direct mapping between a toxin’s primary accumulation sites and the exact organs where tumors most reliably appear. As we laid out in “Why Tumors Form in Specific Organs,” the body does not pick tumor locations at random. It strategically forms them in tissues that are either most heavily burdened by the toxin or best equipped biologically to isolate and contain it — protecting more critical organs in the process.
PFAS (“forever chemicals”) follow this pattern with striking precision:
Liver: The single largest storage depot for long-chain PFAS such as PFOS and PFOA. As the body’s central detoxification hub, the liver becomes overwhelmed; tumors here act as an expanded “second liver” sequestration compartment — directly extending the fructose-handling mechanism we described earlier.
Kidneys: PFAS bind tightly to proteins and undergo extensive reabsorption in the renal tubules, leading to exceptionally high tissue concentrations. This explains the robust epidemiological link between PFAS exposure and kidney cancer.
Lungs: Multiple human tissue studies show lungs often accumulate among the highest PFAS levels of any organ. The resulting chronic burden correlates with increased risk of lung adenocarcinoma, fitting the pattern of airborne or systemic toxins being sequestered locally.
Testes: PFAS readily concentrate in testicular tissue, producing one of the strongest and most consistent associations in the entire PFAS-cancer literature: testicular cancer.
This is no coincidence. In TST, tumors arise exactly where the toxin load is highest — creating localized, high-metabolic sinks (via protein binding and lipid droplets) that pull PFAS out of circulation and isolate them. Just as seed oils map to the prostate due to its lipid-storage biology, or oxalates map to the kidneys due to filtration overload, PFAS map to the liver, kidneys, lungs, and testes because these tissues become the body’s frontline containment zones.
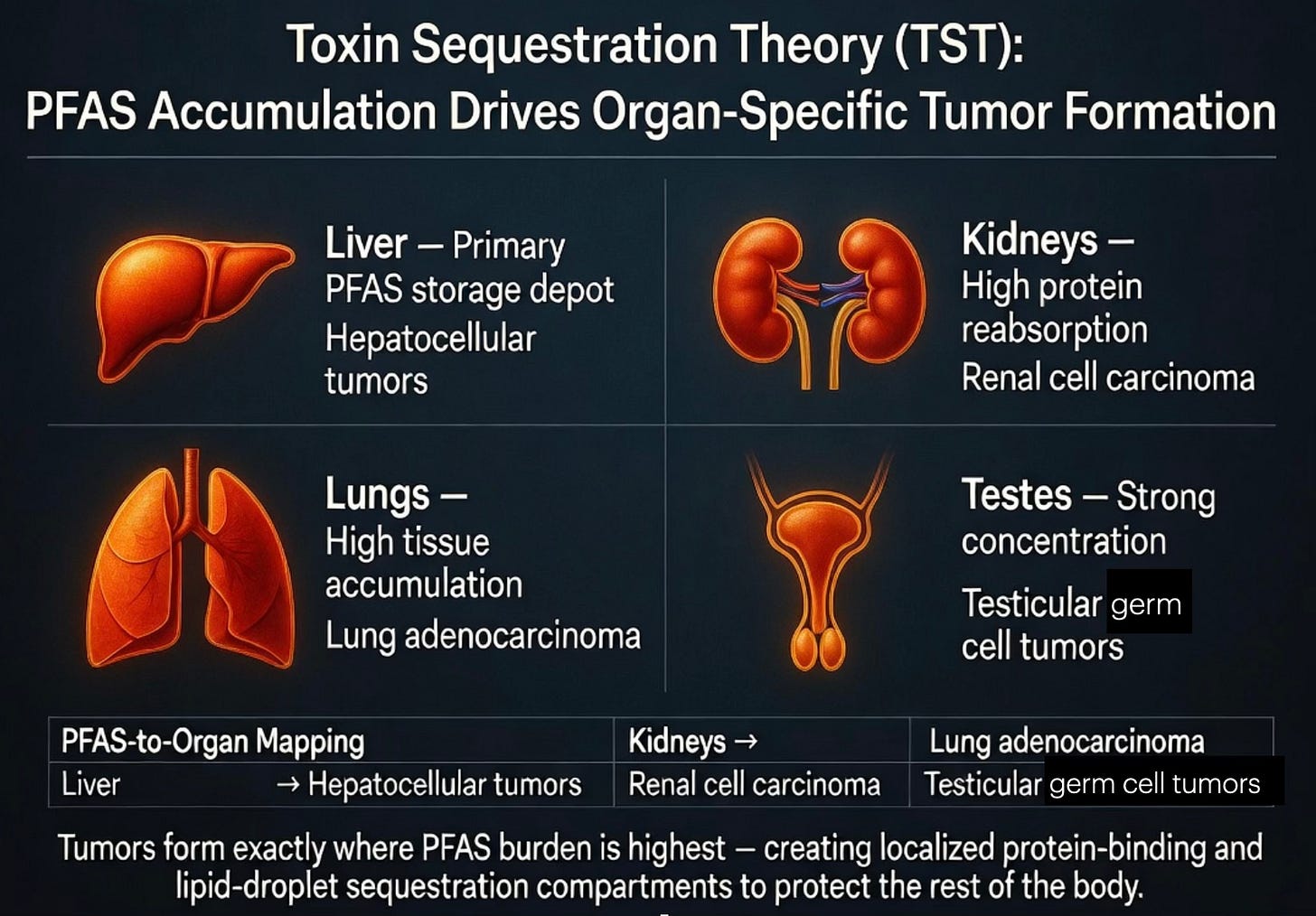
The mapping reinforces the entire series: the location of the tumor reveals the underlying toxin. Reduce the PFAS burden in these specific organs, and the biological pressure to maintain these protective sequestration sites should decline — offering a clear, mechanism-driven path forward.
Practical Implications: Reducing PFAS Burden and Supporting Sequestration
Toxin Sequestration Theory views PFAS-linked cancers as the body’s last-resort effort to isolate “forever chemicals” — just as it sequesters glucose, fructose, and seed oils. Understanding this opens practical steps:
Minimize new exposure
Use PFAS-certified water filters (activated carbon + reverse osmosis).
Ditch non-stick cookware, stain-resistant fabrics, and plastic food packaging where possible.
Choose fresh, unpackaged foods and glass/stainless storage.
Lower incoming load reduces pressure on the sequestration system. As exposure drops, the need for expanded tumor compartments often eases.
Support the body’s efforts
Bolster liver and kidney function with hydration, metabolic health, and anti-inflammatory nutrition. Modulating lipid metabolism and growth signaling can help regulate tumor expansion while clearance occurs.
Hopeful takeaway
By addressing the upstream toxic burden — PFAS, seed oils, excess sugars, and other persistent toxins — we decrease the biological necessity for these protective tumors. This offers a coherent, mechanism-based path for prevention and potential regression, consistent across the entire TST series.
Conclusion: Lightening the Forever Load
PFAS nicely illustrate the core principles of TST. Tumors sequester these “forever chemicals” through protein binding and lipid droplets, precisely in the organs where PFAS accumulate most heavily, protecting the liver, kidneys, lungs, brain, and reproductive tissues from even greater harm. The hopeful message is clear: if we reduce the incoming burden of PFAS and other toxins, the biological pressure to maintain these costly sequestration compartments should decline. By minimizing exposure, supporting natural detox pathways, and addressing the upstream toxic load, we give the body less reason to build or expand these protective vaults.



Thank you for the great article and critical information.
Sigh... only human beings would invent something to poison themselves, and elect people who would still allow it. Geez...