Why Tumors Form in Specific Organs: Tissue-Specific Toxin Sequestration
Why do tumors occur in some organs but not others? Can we map specific toxins onto specific organs for cancer? This post answers these questions from the perspective of Toxin Sequestration Theory.
Welcome to my blog! Before I dive into today’s topic, let me welcome new subscribers, many of whom just joined after watching my interview with Dave Mac. It was wonderful of Dave to have me on his podcast to discuss my new theory of cancer: Toxic Sequestration Theory (TST). If you haven’t seen it, check out that video:
TST postulates that cancer tumors are extra tissue that the body intentionally builds to sequester, store, and process toxins. In previous blog posts, I gave evidence supporting this view for a wide range of toxins: deuterium, ammonia, glucose, fructose, seed oils, oxalate, iron, copper, heavy metals, ethanol, cigarette smoke, estrogen, microplastics, and mycotoxins.
Still, there are lingering questions about TST. One of the most common ones I receive is this: “Why does cancer show up so consistently in certain organs — the breast, prostate, liver, colon, or pancreas — and not others?”
It’s a fair question. After all, we see remarkably consistent patterns: prostate cancer in men, breast cancer in women, liver tumors after chronic alcohol intake, and pancreatic tumors after chronic high sugar intake.
It’s hard to rectify these pattens with the mainstream view that tumors are simply “bad luck” caused by random genetic mutations. In this post, I argue that tumor location is not random. It’s strategic.
Moreover, I try to explain why specific toxins are associated with tumors in specific organs. I propose a “toxin-to-organ mapping”, where the body generates a tumor in a specific organ in response to a particular toxic exposure. (For example, seed oils are mapped to prostate tumors, iron and ethanol are mapped to liver tumors, etc.). This toxin-to-organ mapping may allow us to treat specific cancers — if we know what toxins map onto which organs, we can target those toxins for detoxification.
The Mainstream View
First let’s discuss how mainstream oncology explains tissue-specific cancers. They attribute cancer to the progressive accumulation of genetic mutations. In this model, a cell in a given tissue eventually acquires enough mutations to lose growth control, leading to tumor formation. Organ preference is usually explained by “tissue-specific mutation rates,” local carcinogen exposure, or simple random chance.
While this framework has dominated research for decades, it leaves several observations poorly explained:
It fails to account for the striking consistency of organ preference. Systemic toxins such as seed oils, excess iron, estrogenic compounds, and oxalates circulate throughout the entire body, yet tumors reliably and disproportionately appear in specific organs (particularly prostate, breast, liver, and colon). The mainstream model offers no convincing reason why the same circulating toxins would trigger cancer predominantly in one tissue over another.
It cannot explain why certain organs become reliable “hot spots” even when the overall mutation burden across tissues is comparable. If mutations were truly the primary driver, we would expect a more random or evenly distributed pattern of cancer.
It provides no functional explanation for why the body would repeatedly “choose” metabolically active or detoxification-related organs as the preferred site for tumor formation rather than distributing the damage more evenly.

Tissue-Specific Toxin Sequestration Theory
As a clear contrast, I will now give my views on tissue-specific cancers. We can basically summarize this in four core principles, which I will call Tissue-Specific Toxin Sequestration Theory (TSTST):
The body does not sequester toxins uniformly — it strategically chooses specific tissues as tumor sites.
Tumors often form in tissues that are most heavily burdened by specific toxins.
Tumors also often form in tissues that are best equipped to safely sequester and isolate the toxic load.
Critical organs (such as the heart and spleen) are almost never chosen, as tumors there would immediately threaten survival.
Let’s dive into these core principles.
Tumors Form in Heavily Burdened Tissues
Many tumors arise in the body’s primary detoxification and exposure organs — the tissues that face the highest direct or cumulative toxic load.
The liver, as the main chemical processing plant, becomes heavily burdened by iron overload, alcohol (ethanol), fructose, and mycotoxins. When its detoxification capacity is exceeded, the body forms tumors in the liver as a secondary storage site.
The kidneys, which continuously filter blood, concentrate oxalates, heavy metals, and other waste products. This makes them frequent sites of tumor formation when the filtration burden becomes chronic.
The lungs, as the organ with the largest direct exposure to inhaled toxins (cigarette smoke and airborne pollutants), experience intense local redox stress. Tumors here often represent an attempt to sequester and isolate these directly inhaled toxins.
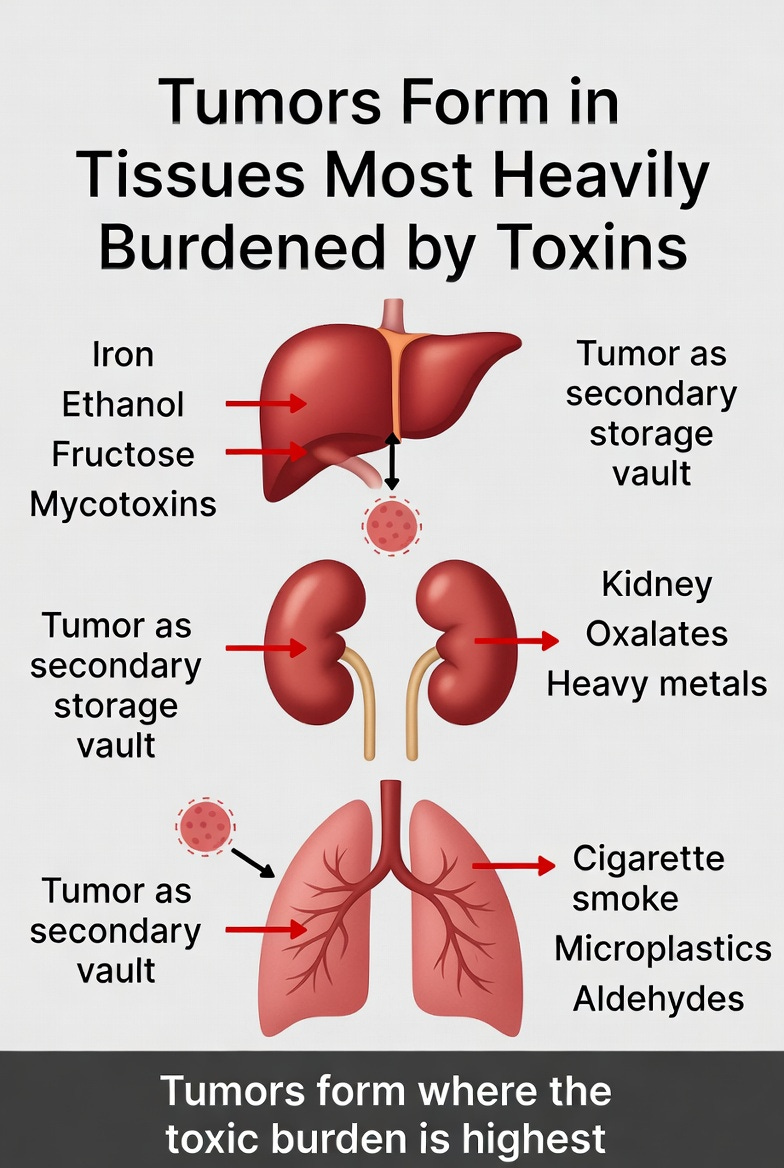
In each case, the tumor is not random. It forms in the organ that is most heavily burdened by the specific toxin the body is struggling to clear.
Tumors Form in Tissues Best Equipped to Safely Sequester Toxins
Sometimes the body chooses a tissue not because it is the most burdened, but because it is best equipped to safely isolate and store the toxin.
Certain organs possess unique biological features that make them effective “vaults”:
The prostate has high levels of fatty acid transporters (such as CD36) and a natural capacity to store lipids in droplets, making it well-suited to sequester the toxic linoleic acid in seed oils.
The breast is highly estrogen-sensitive and has a strong capacity for local estrogen metabolism and uptake. This allows it to act as an effective sequestration site for excess estrogen and its oxidative byproducts.
The colon has a large surface area and direct exposure to gut contents, enabling it to wall off mycotoxins and dietary aldehydes.
In these cases, the tissue is selected because it can better tolerate the formation of a storage site without immediately compromising vital functions. The resulting tumor acts as a localized containment strategy — isolating the toxin so the rest of the body is protected.
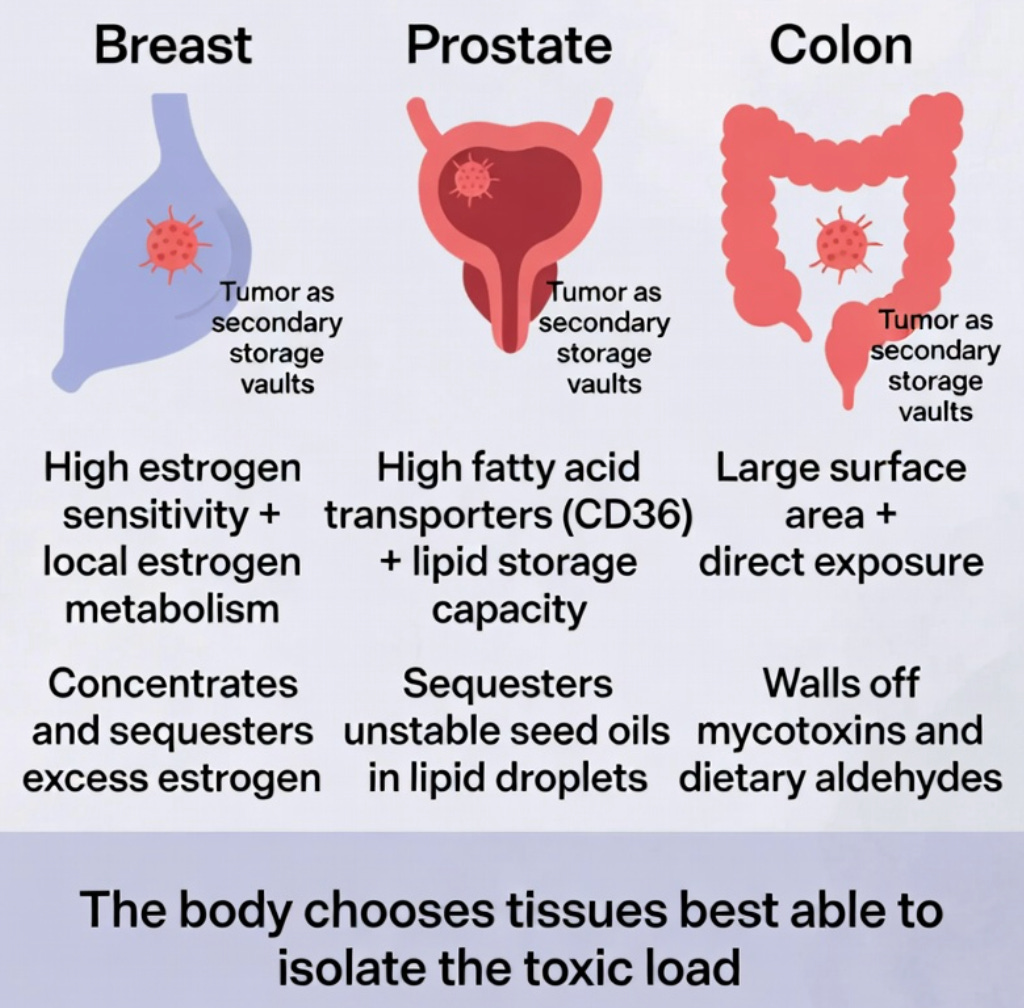
This “best equipped” principle helps explain why some organs become preferred sequestration sites even when they are not the most heavily exposed.
Why the Prostate?
The prostate provides one of the clearest real-world examples of tissue-specific toxin sequestration.
This small gland is not essential for immediate survival, yet it consistently becomes a hotspot for cancer in men. According to my theory (TSTST), this is not random. The prostate is strategically chosen because it is particularly well-equipped to handle one of the most common modern toxins: unstable omega-6 fatty acids from seed oils.
The prostate expresses high levels of fatty acid transporters (especially CD36), allowing it to readily pull linoleic acid out of circulation. It also has a natural capacity to store lipids in droplets. When the body is overloaded with these highly oxidizable fats, the prostate becomes an effective “vault” — isolating the toxic load locally through lipid storage and metabolic adaptation, rather than letting it damage more critical systems.
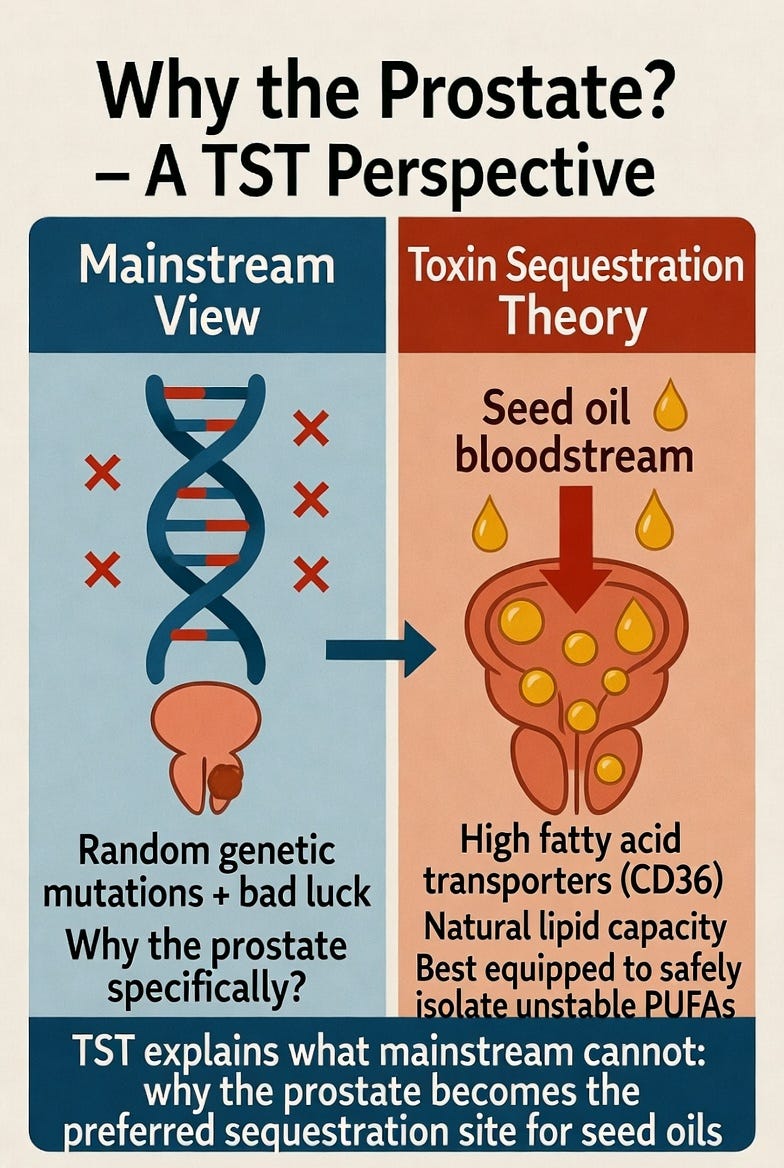
This explains why prostate cancer is so disproportionately common, even though seed oils circulate throughout the entire body. The mainstream mutation model has no convincing answer for this strong tissue preference. TSTST does: the prostate is selected because it is one of the best-equipped tissues to safely sequester and contain unstable PUFAs without immediately threatening survival.
Why Not the Heart?
One of the most telling observations in cancer epidemiology is what doesn’t happen.
Primary tumors in the heart (cardiac cancer) are extraordinarily rare — so rare that many cardiologists go their entire careers without seeing a single case. The same is true for the spleen. These two organs almost never develop primary cancers, even though they are exposed to the same circulating toxins as every other tissue.
From the perspective of TSTST, this is not surprising. It is strategic.
The heart and spleen are critically important for immediate survival. Any significant tumor growth or inflammation in the heart would quickly impair cardiac function and threaten life within days or weeks. Similarly, any significant tumor growth in the spleen would rapidly impair its ability to clear damaged blood cells and fight infections, posing an immediate threat to survival. The body appears to avoid using these vital organs as sequestration sites precisely because forming a tumor there would be too costly.
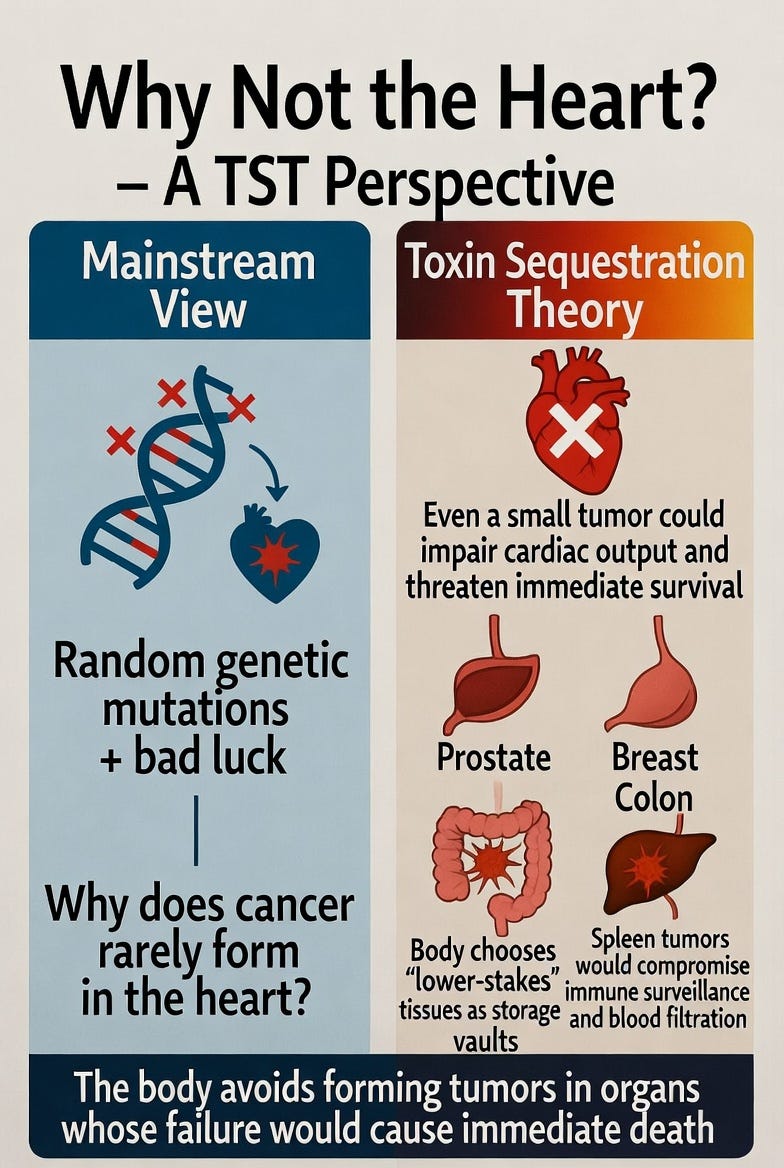
Instead, the body preferentially chooses “lower-stakes” tissues — such as the prostate, breast, colon, or liver — that can better tolerate the formation of a storage vault without immediately compromising survival. These organs can expand, store lipids or metals, and undergo metabolic adaptation with less immediate risk to the organism.
This selective avoidance of critical organs is completely absent from the mainstream mutation model, which would predict tumors should appear randomly across all tissues with similar mutation rates. TST explains it directly: the body is making a calculated decision — it forms tumors where it can safely contain the toxic burden with the least risk to overall survival.
Why specific organs are chosen
With this in mind, we can now see why specific organs are often chosen for cancer tumors. This is summarized in the table below.
We can see that, in some cases, it is a combination of the fact that the organ is heavily burdened and also best able to isolate the toxin. The lung for example is the organ that’s directly exposed to inhaled toxins, receiving a heavy toxic burden. Yet the lung is also capable of walling off particles, so it has a unique ability to protect the body from the toxic effects of inhaled toxins. This could explain why lung cancers are fairly common, ranking as the third most common cancer in the US.
Toxin-to-Organ Mapping: Which Toxins Drive Tumors Where?
We are now in an exciting position to create a toxin-to-organ mapping. The idea is that specific toxins map onto specific organs. This can happen for two key reasons:
The toxin directly impacts that organ (adding to that organ’s toxic burden).
The toxin can be easily sequestered and stored in that organ.
The second reason is especially interesting, as some toxins go to certain organs just because that organ is well equipped to handle that toxin.
For this toxin-to-organ mapping, here are some of the strongest links that appear in the medical literature:
Seed oils → strongest link to Prostate (and also Breast/Colon)
Unstable omega-6 fats are readily incorporated into cell membranes, greatly increasing lipid peroxidation when ROS levels rise.Iron → strongest link to Liver (and Prostate)
Excess iron drives the Fenton reaction, generating highly destructive hydroxyl radicals in iron-rich or iron-accumulating tissues.Excess estrogen → strongest link to Breast
Estrogen-sensitive tissues experience upregulated ROS production through DUOX and NOX enzymes, amplified by copper.Fructose / Glucose → strongest link to Pancreas (and Liver)
Chronic high sugar exposure creates severe glucotoxicity and redox stress in insulin-producing and metabolically active tissues.Oxalates → strongest link to Prostate and Breast (and Kidney)
Calcium oxalate crystals promote local inflammation and oxidative damage in these tissues.Mycotoxins → strongest link to Liver and Colon
These fungal toxins impair mitochondrial function and deplete glutathione, with direct exposure in the gut and heavy processing burden in the liver.Ethanol → strongest link to Liver
As a direct hepatotoxin, ethanol generates high levels of ROS and acetaldehyde, overwhelming the liver’s detoxification capacity.
We can summarize the above information in the figure below, which draws arrows linking specific toxins to specific organs (darker arrows indicate stronger links).
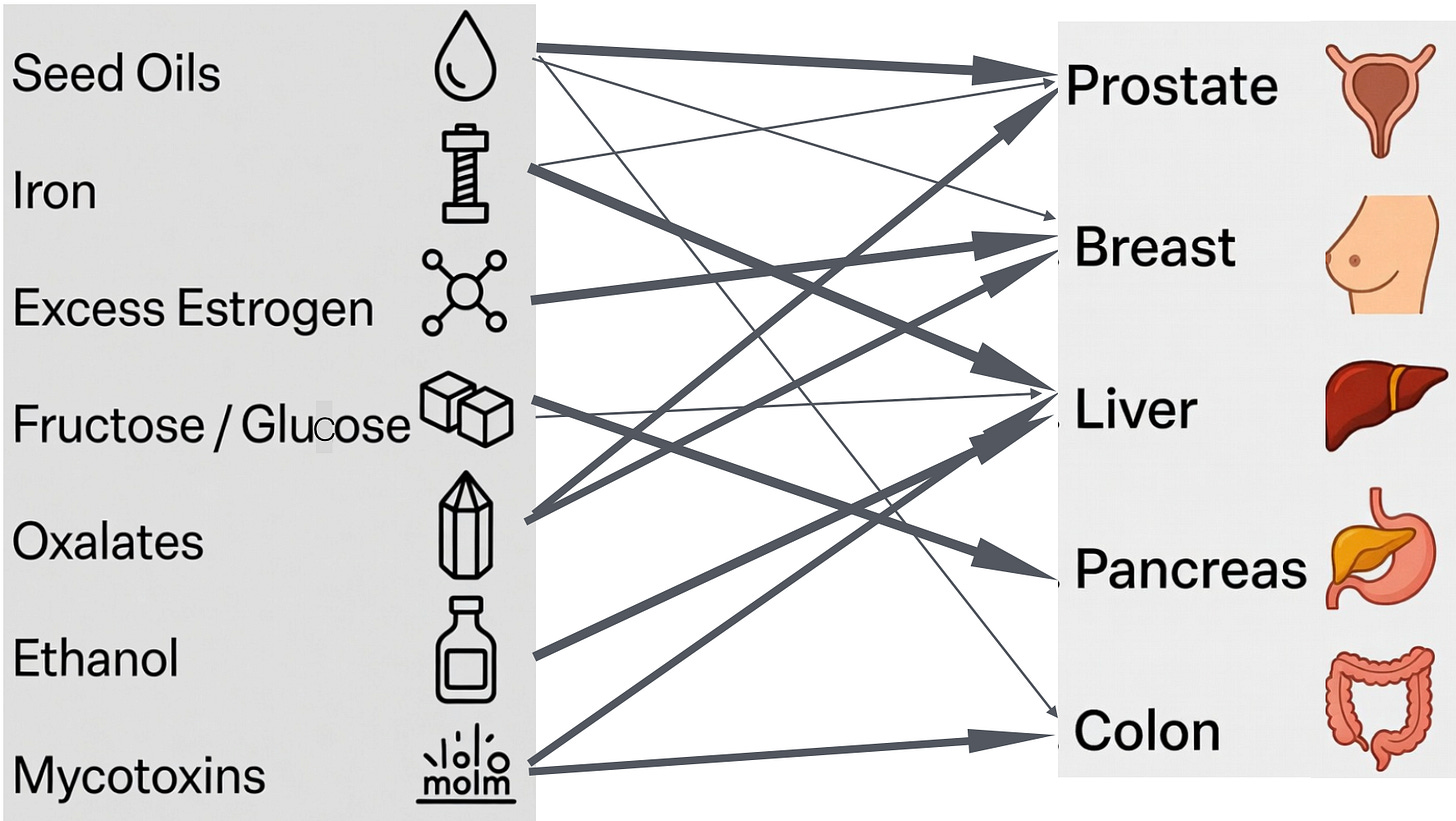
These mappings are not coincidences. Each organ becomes a preferred sequestration site because of its unique biology — its transporters, metabolic activity, storage capacity, and inability to fully clear the toxin through primary detoxification routes.
Why TSTST Offers a Better Explanation
Let’s revisit the mainstream view and discuss why our theory (TSTST) offers a better explanation. The mainstream mutation-centered model fails to explain one of cancer’s most consistent and puzzling features: its strong preference for specific organs.
It attributes tumor location to random genetic mutations or vaguely defined “tissue-specific mutation rates.” According to this view, cancer simply appears wherever enough mutations happen to accumulate. This leaves a critical question completely unanswered: why do the same circulating toxins reliably and disproportionately trigger tumors in certain organs — and not others?
TSTST gives a more coherent and functional answer.
The body does not form tumors randomly. It strategically selects specific tissues that are either most heavily burdened by a particular toxin or best equipped to sequester and safely isolate it without immediately threatening the survival of the organism as a whole.
For example, the mainstream model cannot explain why prostate cancer is so disproportionately common compared to other tissues exposed to the same circulating toxic PUFAs from seed oils. TSTST explains it directly: the prostate is one of the best-equipped tissues to isolate and contain that specific toxin, and it’s also not the most critical organ for immediate survival. When the body is overloaded with unstable seed oils (linoleic acid), the prostate becomes an effective “vault” for sequestering these toxic fats locally, because of its high expression of fatty acid transporters and a natural ability to store lipids in droplets
This principle elegantly accounts for patterns the mutation model cannot: why breast cancer dominates in women, why the liver becomes a hotspot after iron or alcohol overload, and why the pancreas is especially vulnerable to chronic sugar exposure.
By reframing tumors as strategic storage vaults rather than random genetic accidents, our theory offers a clearer, more logical, and ultimately more useful explanation of why cancer appears where it does.
Implications for Treatment
Recognizing tissue-specific toxicity shifts cancer strategy from attacking tumors to reducing the upstream toxic load. Prevention becomes targeted: lower the dominant toxins for each organ — seed oils for prostate, excess estrogen for breast, iron for liver, fructose for pancreas, and so on. For existing tumors, aggressive removal or metabolic starvation may be counterproductive if the driving toxins remain high. Instead, the priority should be lightening the overall redox burden while supporting antioxidant defenses. Early intervention offers the best chance. When the toxic load is reduced before extensive damage occurs, the body may no longer need the tumor as a sequestration vault. The goal is simple: remove the reason the body created the tumor in the first place.
Conclusion
I think this tissue-specific version of my cancer theory is pretty exciting! It gives a plausible explanation for why tumors always seem to appear in the same tissues (like prostate, breast, liver, colon, lung) and don’t appear in other tissues (like heart, spleen). Moreover, the toxin-to-organ mapping gives us an actionable way to treat specific cancers. If someone has breast cancer, we can advise them to reduce their excess estrogen, seed oils, and oxalates — whereas if someone has liver cancer, we can advise them to reduce their excess iron, ethanol, and fructose.
Please let me know your thoughts and feedback on these ideas. Thanks for reading!





Interesting framework — the idea of tissue-specific vulnerability is something I’ve been thinking about as well.
Curious how this would account for metastasis, and how we can actually verify toxin or sequestration levels in tissues?
Yes having toxicology reports on cancer tumors would be important information.